Key Words: Tranexamic acid, epistaxis, Epistemonikos, GRADE
Abstract
INTRODUCTION
Spontaneous epistaxis is one of the most frequent problems in emergency services. New treatment alternatives have emerged, including topical tranexamic acid. However, there is controversy about the actual efficacy of this alternative.
METHODS
We searched in Epistemonikos, the largest database of systematic reviews in health, which is maintained by screening multiple information sources, including MEDLINE, EMBASE, Cochrane, among others. We extracted data from the systematic reviews, reanalyzed data of primary studies, conducted a meta-analysis and generated a summary of findings table using the GRADE approach.
RESULTS AND CONCLUSIONS
We identified five systematic reviews that analyzed only one primary study, corresponding to a randomized trial. We concluded it is not clear whether topical tranexamic acid has any impact on hemostasis or risk of rebleeding because the certainty of the evidence is very low. On the other hand, its use could increase adverse effects.
Problem
Epistaxis is one of the most frequent problems in emergency services. It can result in significant blood loss, particularly in anticoagulant treatment users or in people with comorbidities [1]. The most common approach is cauterization of the bleeding vessel. When the bleeding site is not visible, nasal plugs are used, but they generate discomfort [2], so new therapeutic alternatives are actively sought.
Among many potential treatments for this condition, is tranexamic acid, an antifibrinolytic agent which has proven effective in reducing bleeding in different surgeries, trauma and non-surgical diseases [1], [3]. However, the efficacy and safety of topical tranexamic acid in spontaneous epistaxis is unclear
Methods
We searched in Epistemonikos, the largest database of systematic reviews in health, which is maintained by screening multiple information sources, including MEDLINE, EMBASE, Cochrane, among others, to identify systematic reviews and their included primary studies. We extracted data from the identified reviews and reanalyzed data from primary studies included in those reviews. With this information, we generated a structured summary denominated FRISBEE (Friendly Summary of Body of Evidence using Epistemonikos) using a pre-established format, which includes key messages, a summary of the body of evidence (presented as an evidence matrix in Epistemonikos), meta-analysis of the total of studies when it is possible, a summary of findings table following the GRADE approach and a table of other considerations for decision-making.
|
Key messages
|
About the body of evidence for this question
|
What is the evidence. |
We found five systematic reviews [1], [3], [4], [5], [6], which included only one primary study that answered the question of interest [2], corresponding to a randomized trial. This table and the summary in general are based on this trial [2]. |
|
What types of patients were included* |
The trial [2] included patients aged 18 years or older, with spontaneous epistaxis, coming from both upper, posterior and/or Kiesselbach area of the nasal septum. Patients with a fracture of the skull or nose and perforation of the nasal septum were excluded, as well as patients with a history of hemostasis alteration. |
|
What types of interventions were included* |
The trial [2] compared the use of 10% tranexamic acid gel (15 ml) against a glycine gel. After the application, both groups received a piece of cotton that they had to keep for 30 minutes in the nostril. |
|
What types of outcomes |
The trial reported multiple outcomes, which were presented by the different systematic reviews as follows:
The follow-up of the trial was 10 days. |
* The information about primary studies is extracted from the systematic reviews identified, unless otherwise specified.
Summary of findings
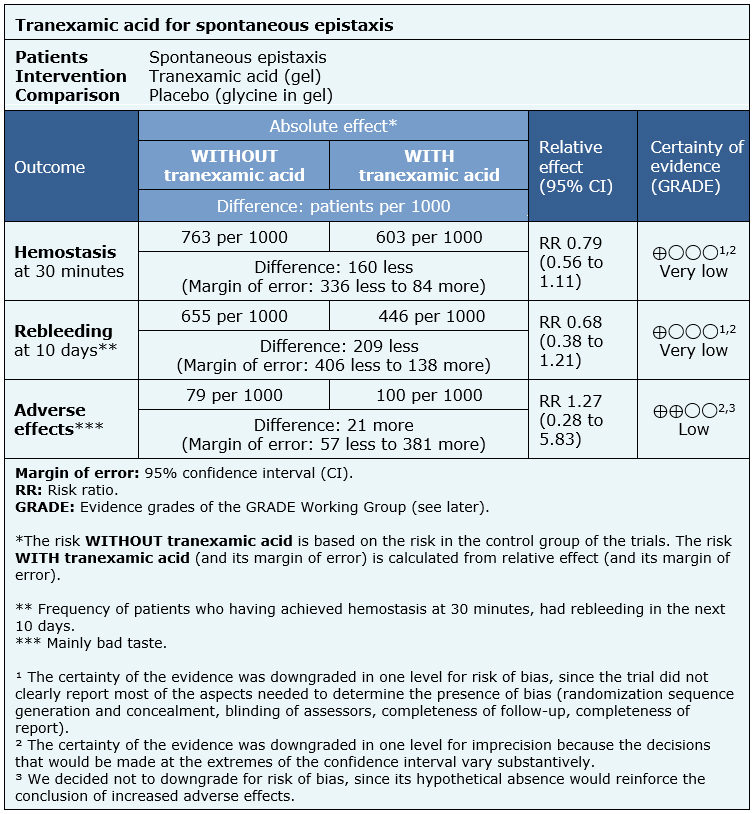
The information on the effects of topical tranexamic acid is based on only one randomized trial that included 68 patients [2].
The trial reported hemostasis at 30 minutes (68 patients), rebleeding at 10 days (47 patients) and adverse effects (68 patients).
The summary of findings is the following:
- It is not clear whether the use of topical tranexamic acid impacts haemostasis because the certainty of the evidence is very low.
- It is not clear whether the use of topical tranexamic acid decreases rebleeding because the certainty of the evidence is very low.
- The use of tranexamic acid might increase adverse effects, but the certainty of the evidence is low.
| Follow the link to access the interactive version of this table (Interactive Summary of Findings – iSoF) |
Other considerations for decision-making
|
To whom this evidence does and does not apply |
|
| About the outcomes included in this summary |
|
| Balance between benefits and risks, and certainty of the evidence |
|
| Resource considerations |
|
| What would patients and their doctors think about this intervention |
|
|
Differences between this summary and other sources |
|
| Could this evidence change in the future? |
|
How we conducted this summary
Using automated and collaborative means, we compiled all the relevant evidence for the question of interest and we present it as a matrix of evidence.

Follow the link to access the interactive version: Tranexamic acid for the management of spontaneous epistaxis.
Notes
The upper portion of the matrix of evidence will display a warning of “new evidence” if new systematic reviews are published after the publication of this summary. Even though the project considers the periodical update of these summaries, users are invited to comment in Medwave or to contact the authors through email if they find new evidence and the summary should be updated earlier.
After creating an account in Epistemonikos, users will be able to save the matrixes and to receive automated notifications any time new evidence potentially relevant for the question appears.
This article is part of the Epistemonikos Evidence Synthesis project. It is elaborated with a pre-established methodology, following rigorous methodological standards and internal peer review process. Each of these articles corresponds to a summary, denominated FRISBEE (Friendly Summary of Body of Evidence using Epistemonikos), whose main objective is to synthesize the body of evidence for a specific question, with a friendly format to clinical professionals. Its main resources are based on the evidence matrix of Epistemonikos and analysis of results using GRADE methodology. Further details of the methods for developing this FRISBEE are described here (http://dx.doi.org/10.5867/medwave.2014.06.5997)
Epistemonikos foundation is a non-for-profit organization aiming to bring information closer to health decision-makers with technology. Its main development is Epistemonikos database (www.epistemonikos.org).
Potential conflicts of interest
The authors do not have relevant interests to declare.
 Esta obra de Medwave está bajo una licencia Creative Commons Atribución-NoComercial 3.0 Unported. Esta licencia permite el uso, distribución y reproducción del artículo en cualquier medio, siempre y cuando se otorgue el crédito correspondiente al autor del artículo y al medio en que se publica, en este caso, Medwave.
Esta obra de Medwave está bajo una licencia Creative Commons Atribución-NoComercial 3.0 Unported. Esta licencia permite el uso, distribución y reproducción del artículo en cualquier medio, siempre y cuando se otorgue el crédito correspondiente al autor del artículo y al medio en que se publica, en este caso, Medwave.

INTRODUCCIÓN
La epistaxis espontánea es uno de los problemas más frecuentes en consultas de urgencia. Existen nuevas alternativas de tratamiento, entre las que se encuentra el ácido tranexámico tópico. Sin embargo, su rol en el manejo de la epistaxis espontánea sigue siendo poco claro, existiendo controversia en cuanto a su efectividad y seguridad.
MÉTODOS
Realizamos una búsqueda en Epistemonikos, la mayor base de datos de revisiones sistemáticas en salud, la cual es mantenida mediante el cribado de múltiples fuentes de información, incluyendo MEDLINE, EMBASE, Cochrane, entre otras. Extrajimos los datos desde las revisiones identificadas, analizamos los datos de los estudios primarios, realizamos un metanálisis y preparamos una tabla de resumen de los resultados utilizando el método GRADE.
RESULTADOS Y CONCLUSIONES
Identificamos cinco revisiones sistemáticas que en conjunto incluyeron un estudio primario, el cual corresponde a un ensayo aleatorizado. Concluimos que no está claro si el uso de ácido tranexámico tópico impacta en hemostasia o resangrado porque la certeza de la evidencia es muy baja, además su uso podría aumentar los efectos adversos.
 Authors:
María Jesús Santander[1,2], Andrés Rosenbaum[2,3], Matías Winter[2,3]
Authors:
María Jesús Santander[1,2], Andrés Rosenbaum[2,3], Matías Winter[2,3]
Affiliation:
[1] Facultad de Medicina, Pontificia Universidad Católica de Chile, Santiago, Chile
[2] Proyecto Epistemonikos, Santiago, Chile
[3] Departamento de Otorrinolaringología, Facultad de Medicina, Pontificia Universidad Católica de Chile, Santiago, Chile
E-mail: mwinterd@gmail.com
Author address:
[1] Centro Evidencia UC Pontificia Universidad Católica de Chile Diagonal Paraguay 476 Santiago Chile

Citation: Santander M, Rosenbaum A, Winter M. Topical tranexamic acid for spontaneous epistaxis. Medwave 2018;18(8):e7371 doi: 10.5867/medwave.2018.08.7371
Submission date: 15/11/2018
Acceptance date: 29/11/2018
Publication date: 10/12/2018
Origin: This article is a product of the Evidence Synthesis Project of Epistemonikos Fundation, in collaboration with Medwave for its publication.
Type of review: Non-blinded peer review by members of the methodological team of Epistemonikos Evidence Synthesis Project.

Comments (0)
We are pleased to have your comment on one of our articles. Your comment will be published as soon as it is posted. However, Medwave reserves the right to remove it later if the editors consider your comment to be: offensive in some sense, irrelevant, trivial, contains grammatical mistakes, contains political harangues, appears to be advertising, contains data from a particular person or suggests the need for changes in practice in terms of diagnostic, preventive or therapeutic interventions, if that evidence has not previously been published in a peer-reviewed journal.
No comments on this article.
To comment please log in
 Medwave provides HTML and PDF download counts as well as other harvested interaction metrics.
Medwave provides HTML and PDF download counts as well as other harvested interaction metrics. There may be a 48-hour delay for most recent metrics to be posted.

- Kamhieh Y, Fox H. Tranexamic acid in epistaxis: a systematic review. Clinical Otolaryngology. 2016;41(6):771-776.
- Tibbelin A, Aust R, Bende M, Holgersson M, Petruson B, Rundcrantz H et al. Effect of Local Tranexamic Acid Gel in the Treatment of Epistaxis. ORL. 1995;57(4):207-209.
- Montroy J, Hutton B, Moodley P, Fergusson NA, Cheng W, Tinmouth A, Lavallée LT, Fergusson DA, Breau RH. The efficacy and safety of topical tranexamic acid: A systematic review and meta-analysis. Transfus Med Rev. 2018; 32(3):165-178.
- Williams A, Biffen A, Pilkington N, Arrick L, Williams RJ, Smith ME, Smith M, Birchall J. Haematological factors in the management of adult epistaxis: systematic review. J Laryngol Otol. 2017 Dec;131(12):1093-1107.
- Musgrave KM, Powell J. A systematic review of anti-thrombotic therapy in epistaxis. Rhinology. 2016 Dec 1;54(4):292-391.
- Ker K, Beecher D, Roberts I. Topical application of tranexamic acid for the reduction of bleeding. Cochrane Database Syst Rev. 2013 Jul 23;(7):CD010562.
- Gary Peksa, Michael Gottlieb. Efficacy of tranexamic acid for the management of epistaxis. PROSPERO 2018 CRD42018084158.
- Wendy Ingram.Novel use of TXA to reduce the need for nasal packing in epistaxis. ICTRP 2018. ISRCTN34153772.
 Kamhieh Y, Fox H. Tranexamic acid in epistaxis: a systematic review. Clinical Otolaryngology. 2016;41(6):771-776.
Kamhieh Y, Fox H. Tranexamic acid in epistaxis: a systematic review. Clinical Otolaryngology. 2016;41(6):771-776.  Tibbelin A, Aust R, Bende M, Holgersson M, Petruson B, Rundcrantz H et al. Effect of Local Tranexamic Acid Gel in the Treatment of Epistaxis. ORL. 1995;57(4):207-209.
Tibbelin A, Aust R, Bende M, Holgersson M, Petruson B, Rundcrantz H et al. Effect of Local Tranexamic Acid Gel in the Treatment of Epistaxis. ORL. 1995;57(4):207-209.  Montroy J, Hutton B, Moodley P, Fergusson NA, Cheng W, Tinmouth A, Lavallée
LT, Fergusson DA, Breau RH. The efficacy and safety of topical tranexamic acid: A
systematic review and meta-analysis. Transfus Med Rev. 2018; 32(3):165-178.
Montroy J, Hutton B, Moodley P, Fergusson NA, Cheng W, Tinmouth A, Lavallée
LT, Fergusson DA, Breau RH. The efficacy and safety of topical tranexamic acid: A
systematic review and meta-analysis. Transfus Med Rev. 2018; 32(3):165-178.  Williams A, Biffen A, Pilkington N, Arrick L, Williams RJ, Smith ME, Smith M,
Birchall J. Haematological factors in the management of adult epistaxis:
systematic review. J Laryngol Otol. 2017 Dec;131(12):1093-1107.
Williams A, Biffen A, Pilkington N, Arrick L, Williams RJ, Smith ME, Smith M,
Birchall J. Haematological factors in the management of adult epistaxis:
systematic review. J Laryngol Otol. 2017 Dec;131(12):1093-1107.  Musgrave KM, Powell J. A systematic review of anti-thrombotic therapy in epistaxis. Rhinology. 2016 Dec 1;54(4):292-391.
Musgrave KM, Powell J. A systematic review of anti-thrombotic therapy in epistaxis. Rhinology. 2016 Dec 1;54(4):292-391.  Ker K, Beecher D, Roberts I. Topical application of tranexamic acid for the reduction of bleeding. Cochrane Database Syst Rev. 2013 Jul 23;(7):CD010562.
Ker K, Beecher D, Roberts I. Topical application of tranexamic acid for the reduction of bleeding. Cochrane Database Syst Rev. 2013 Jul 23;(7):CD010562.  Gary Peksa, Michael Gottlieb. Efficacy of tranexamic acid for the management of epistaxis. PROSPERO 2018 CRD42018084158.
Gary Peksa, Michael Gottlieb. Efficacy of tranexamic acid for the management of epistaxis. PROSPERO 2018 CRD42018084158.  Wendy Ingram.Novel use of TXA to reduce the need for nasal packing in epistaxis. ICTRP 2018. ISRCTN34153772.
Wendy Ingram.Novel use of TXA to reduce the need for nasal packing in epistaxis. ICTRP 2018. ISRCTN34153772. Systematization of initiatives in sexual and reproductive health about good practices criteria in response to the COVID-19 pandemic in primary health care in Chile
Clinical, psychological, social, and family characterization of suicidal behavior in Chilean adolescents: a multiple correspondence analysis








